- Patients Patients
Reproductive Genetics Testing
Patient Resources
Cost & Billing
- Providers Providers
- Genetic Counseling
- Login Login
- Estimate My Cost
Advanced Diagnostic Testing
Reveal® SNP Microarray
With more than 11 years of experience in microarray testing, Labcorp has performed over 230,000 microarrays to date resulting in a robust database to support variant interpretation.
Labcorp’s Reveal SNP microarray has more than 2.6 million genomic markers and is constructed with dense probe coverage in regions of known genetic function to maximize detection of pathogenic variants. The array uses a combination of more than 1.9 million region-specific structural probes to detect copy number changes and ~750,000 SNP probes to detect copy number changes, copy neutral changes, triploidy and molar genotypes. Reveal SNP microarray has applications for pediatric (cord blood, peripheral blood, and buccal swab), prenatal (amniotic fluid and chorionic villus), and POC (fresh tissue and formalin-fixed paraffin-embedded) sample types utilizing the same array for all applications. This enables the reanalysis of data after delivery using pediatric cut-offs without the need for additional specimen, as well as the ability to apply findings between specimen types.
When a clinical phenotype does not fit a specific syndrome, microarray analysis can identify the etiology. Reveal will identify and characterize abnormalities currently detected by routine cytogenetics (with the exception of balanced chromosome rearrangements) as well as genetic changes that may be missed by standard karyotype.
Reveal SNP microarray - Prenatal
Detects genomic imbalances related to congenital abnormalities, intellectual disabilities, miscarriage, and stillbirth. Can detect copy neutral changes associated with increased risk for autosomal recessive conditions, uniparental disomy (UPD), identity by descent (IBD) and molar genotypes.
Reveal SNP microarray - Pediatric
Enhances the detection of chromosome abnormalities. Clinical Indications: Individuals with nonsyndromic congenital anomalies, dysmorphic features, developmental delay, intellectual disability, and/or autism spectrum disorders (ASD).
Reveal SNP microarray - POC
Detects chromosomal imbalances related to fetal loss. Can be performed directly on POC tissue without having to grow cells, optimizing success rate and turnaround time.
Comprehensive test menu and flexible testing options
Labcorp can perform familial studies following an abnormal result via qPCR, chromosome analysis, FISH or microarray. Following an abnormal POC, we are also able to test chorionic villi or amniotic fluid samples, as well as cord blood samples in the nursery to evaluate future at-risk pregnancies and newborn.
Reveal can be ordered as a standalone test or it can run as a reflex to or concurrently with other tests, including FISH and chromosome analysis. Below is an example of a Reveal ordering pathway:
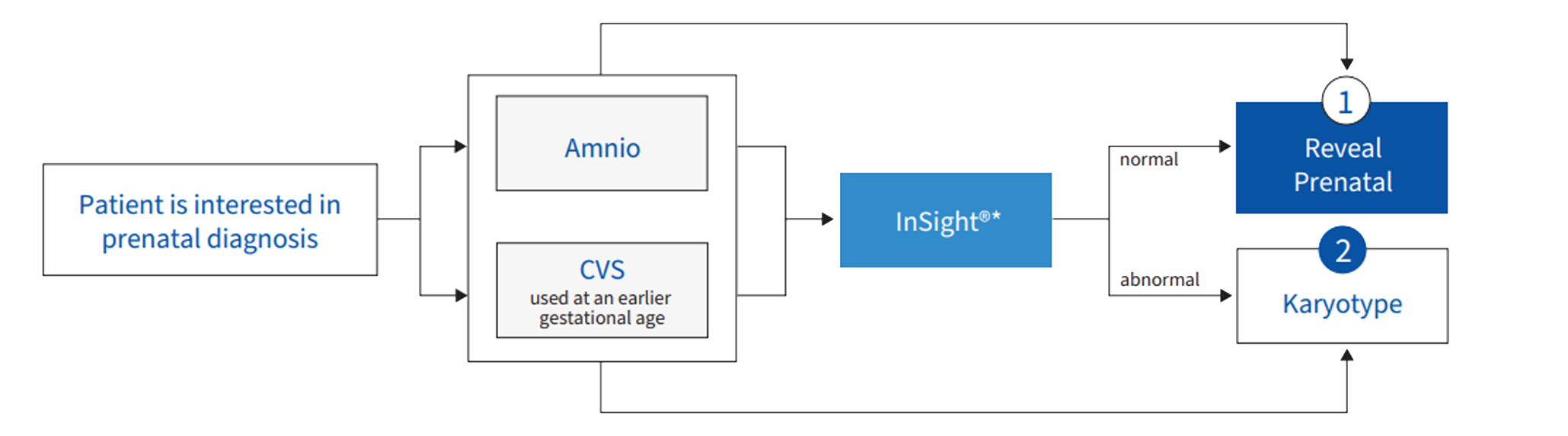

Other testing options
In addition to carrier screening, we also offer: serum screening and noninvasive prenatal screening (NIPS, sometimes called NIPT or cell-free DNA), including the pioneering whole-genome NIPS; diagnostic testing that can be used on prenatal and products of conception (POC) samples, as well as pediatric, cord blood or adult samples; and hereditary cancer screening.

Navigating the Genetic Complexity of Autism Spectrum Disorder (ASD)
Determining the etiology of autism spectrum disorder can help support appropriate condition management and the provision of a realistic diagnosis for the nearly 1 in 36 children diagnosed with autism.1 As such, professional societies recommend SNP microarray as a first-line test for pediatric patients presenting with symptoms of ASD.2-4 Labcorp’s recently enhanced Reveal® SNP Microarray Pediatric provides a high-resolution, genome-wide analysis to identify copy number and copy neutral abnormalities, giving you deep diagnostic insights.
Additional Resources
Understanding Costs
Use our cost estimator get an immediate estimate for most tests we offer based on your specific insurance plan (so have your policy number handy).
Understand Your Results
If you have already had a test, we can answer questions about your results.
Genetic Counseling
Genetic counselors translate and communicate genetic information into practical, understandable terms.
References
- Maenner MJ, Warren Z, Williams AR, et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years - Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill Summ. 2023;72(2):1-14. Published 2023 Mar 24. doi:10.15585/mmwr.ss7202a1
- Manning M, Hudgins L. Professional Practice and Guidelines Committee. Array-based technology and recommendations for utilization in medical genetics practice for detection of chromosomal abnormalities. Genet Med. 2010;12(11):742-745. doi:10.1097/GIM.0b013e3181f8baad
- Miller DT, Adam MP, Aradhya S, et al. Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet. 2010;86(5):749-764. doi:10.1016/j.ajhg.2010.04.006
- Shen Y, Dies KA, Holm IA, et al. Clinical genetic testing for patients with autism spectrum disorders. Pediatrics. 2010;125(4):e727-e735. doi:10.1542/peds.2009-1684



